Why Curves Progress After Spinal Fusion: The Adjacent Segment Question
The surgery worked. The fused segments are straight. The X-ray confirmed it.
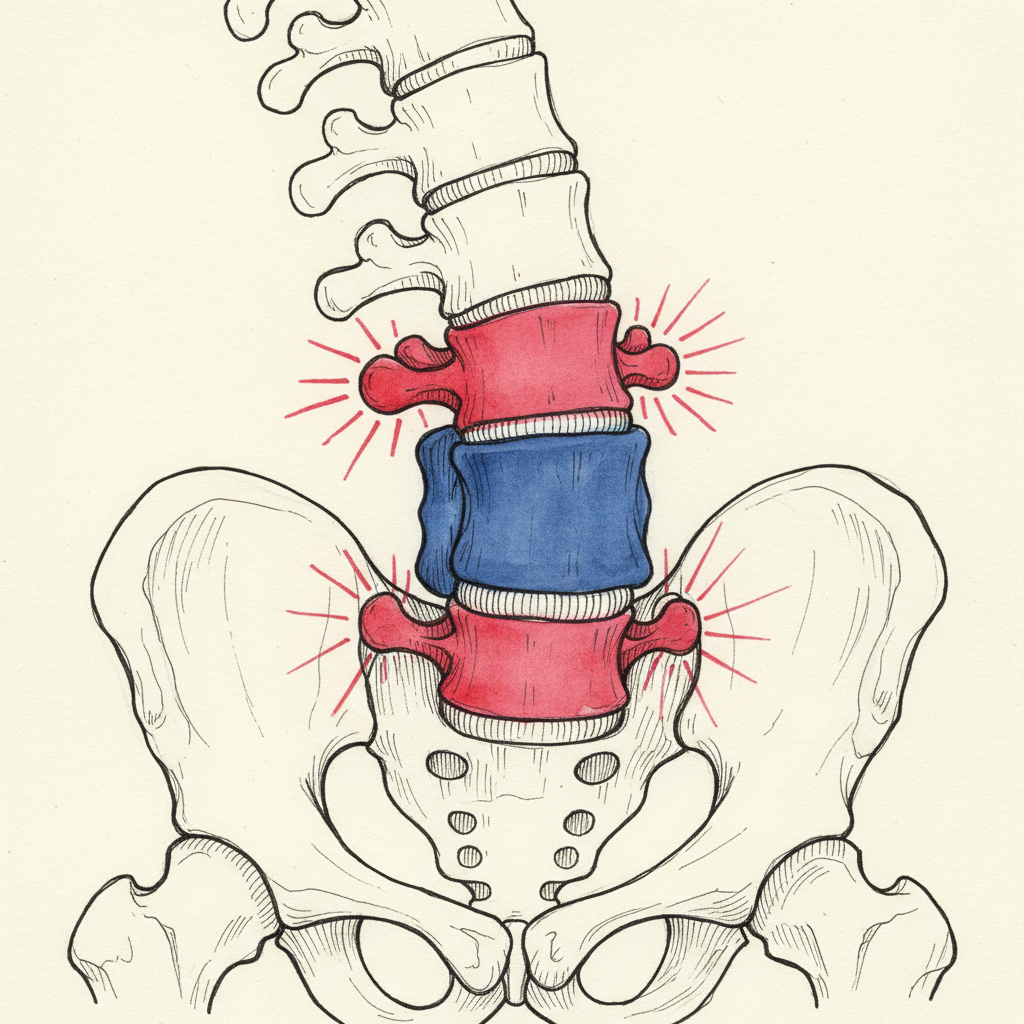
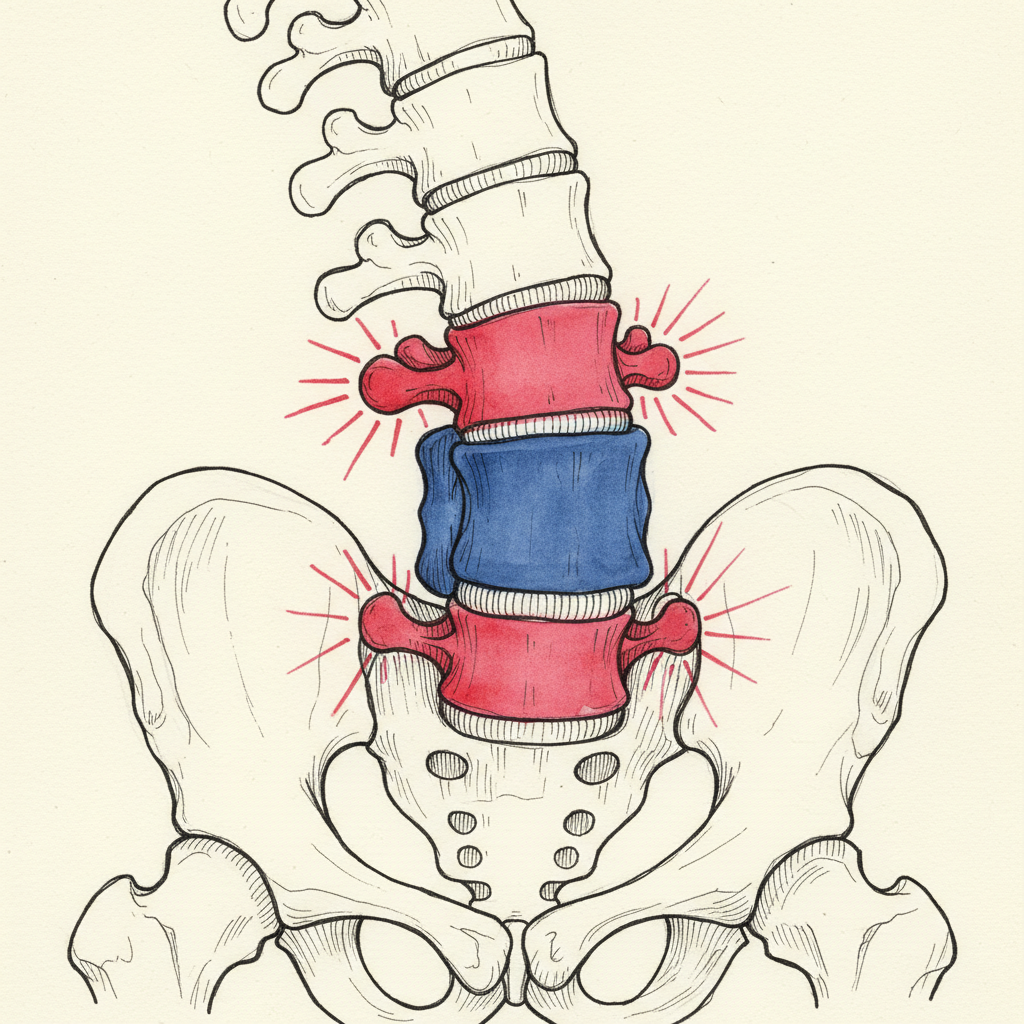
Three years later, the segment above the fusion is degenerating. Five years later, there is a new curve developing where there was no curve before. The surgeon said the fusion would stabilize the spine. The fused segments are stable. Everything around them is not.
This is not a failure of the surgery. It is a documented phenomenon with a name, a mechanism, and an incidence rate that every person considering spinal fusion deserves to know before they consent.
It is called adjacent segment disease. And the standard explanation for it is real but incomplete.
Medical Disclaimer: This article is educational. It does not constitute medical advice. Spinal fusion is a legitimate, sometimes necessary surgical intervention. If you are considering surgery or have already had surgery, work with your surgical team. Nothing here replaces that relationship. What follows is a research summary and a hypothesis about contributing mechanisms that are not yet part of the standard surgical conversation.
What Happens at the Segments Above and Below a Fusion
Spinal fusion eliminates motion at the fused vertebral levels. That is its purpose. The segments are locked together with hardware, bone graft, or both. The curve at those levels is mechanically prevented from progressing.
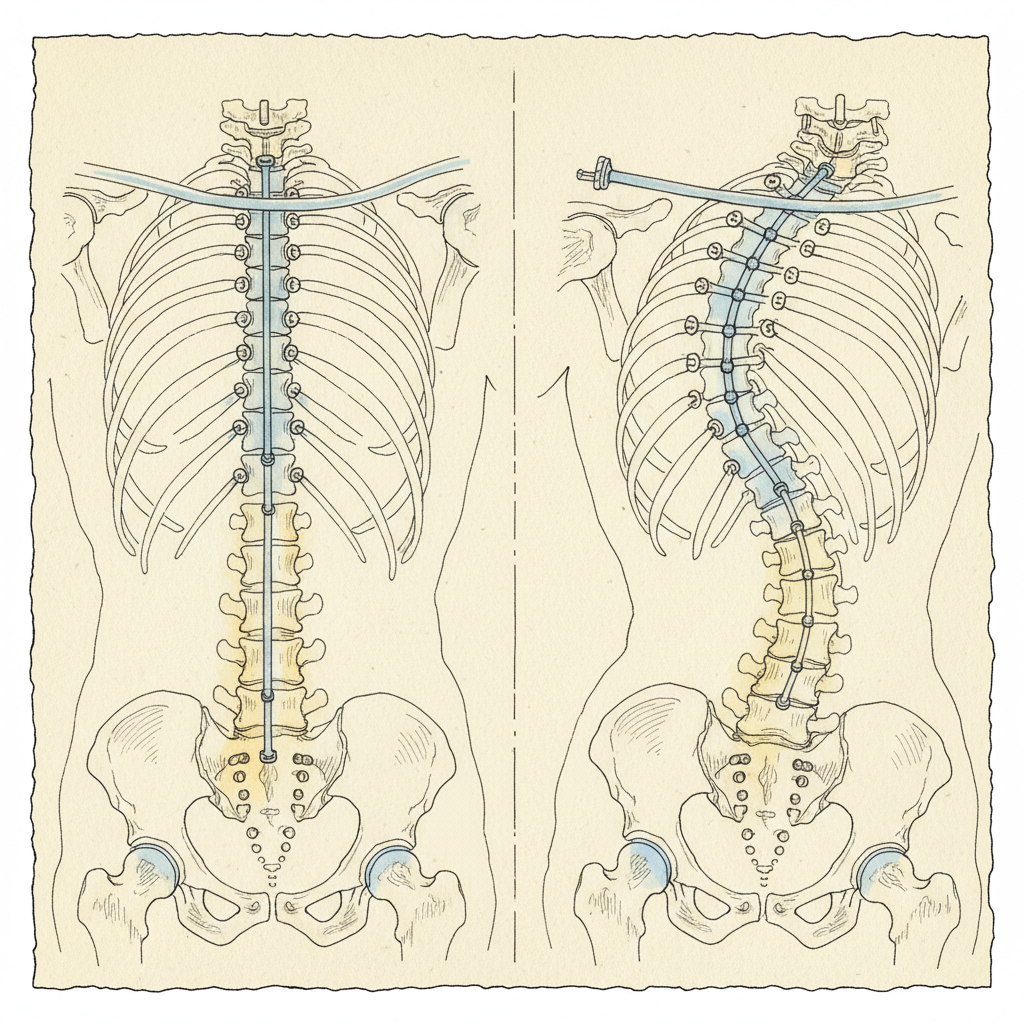
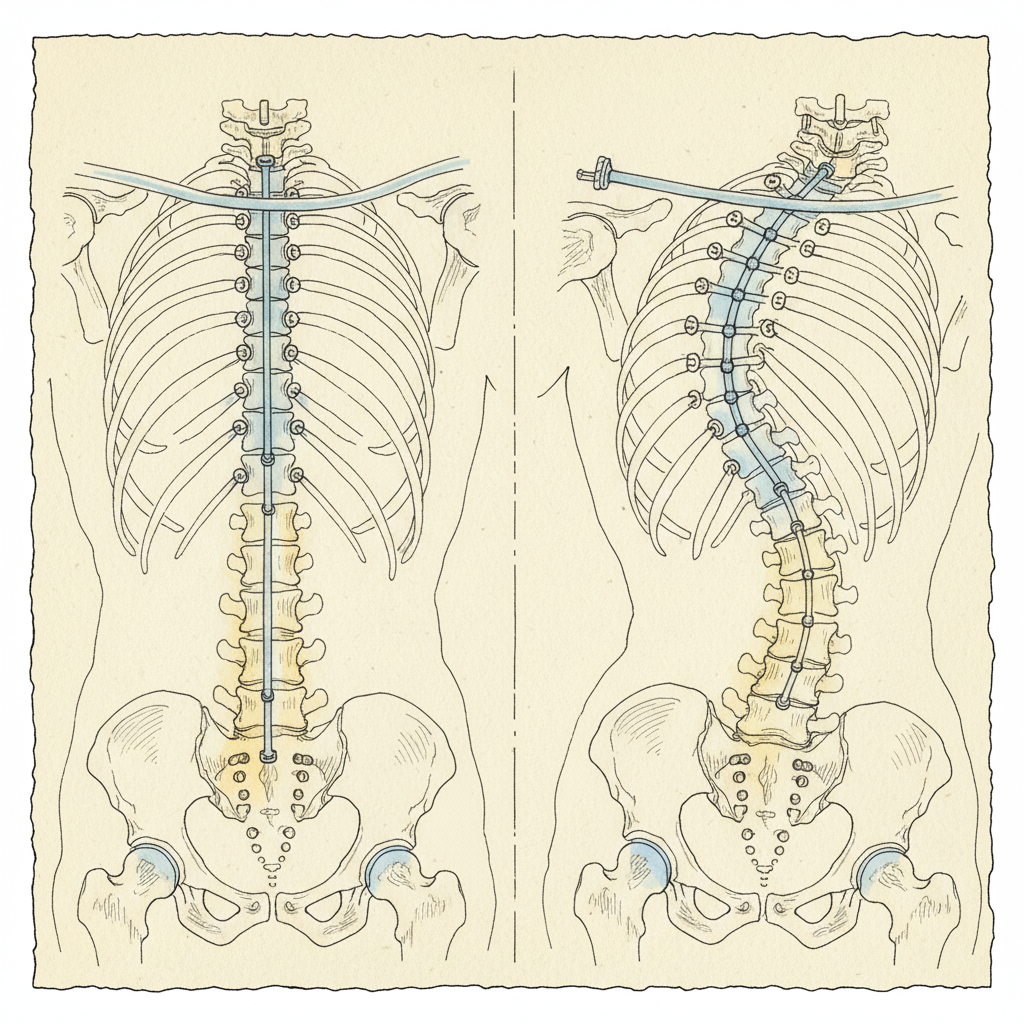
The segments immediately above and below the fusion are now the transition zones. They border a rigid structure on one side and a mobile spine on the other. They bear loads they were not designed to carry alone.
Over time, those transition segments show measurable degeneration. Disc height loss. Facet joint arthropathy. Ligamentous laxity. New curve development. New stenosis. This is adjacent segment disease.
The research is unambiguous.
Helgeson and colleagues published a systematic review in 2010 analyzing the incidence of adjacent segment disease following lumbar and thoracolumbar fusion. Radiographic adjacent segment degeneration, meaning changes visible on imaging whether or not the patient has symptoms, appeared in 25 to 60 percent of patients at ten or more years after surgery (Helgeson et al., 2010). Ghiselli and colleagues found similar numbers in their 10-year follow-up: adjacent segment disease developed in a significant proportion of patients following lumbar fusion (Ghiselli et al., 2004). Cheh and colleagues tracked long-term outcomes after lumbar fusion and documented radiographic adjacent segment degeneration in approximately one-third of patients, with symptomatic disease requiring additional surgery in a substantial subset (Cheh et al., 2007).
Symptomatic adjacent segment disease, meaning degeneration that produces pain or neurological symptoms severe enough to warrant clinical attention, occurs in an estimated 5 to 25 percent of patients depending on the study, the follow-up period, and how symptoms are defined (Helgeson et al., 2010; Cheh et al., 2007).
These are not outlier findings. They are the documented norm for long-term follow-up after spinal fusion.
Adjacent segment disease (ASD) is the development of new degeneration, instability, or deformity at the spinal levels immediately above or below a surgical fusion. It is the most common long-term complication following spinal fusion. Helgeson et al. (2010) conducted a systematic review and found radiographic evidence of adjacent segment degeneration in 25-60% of patients at 10+ years post-fusion. Ghiselli et al. (2004) reported similar findings in a 10-year follow-up of lumbar fusion patients. Cheh et al. (2007) documented that approximately one-third of patients developed radiographic ASD after lumbar fusion, with a significant subset requiring revision surgery. Symptomatic ASD, which produces pain or neurological symptoms requiring clinical intervention, occurs in 5-25% of patients depending on follow-up duration and diagnostic criteria. The standard mechanical explanation attributes ASD to increased loading at the transition segments after motion is eliminated at the fused levels.
Proximal Junctional Kyphosis: The Most Common Form
The most clinically significant form of adjacent segment disease in adult deformity surgery has its own name. Proximal junctional kyphosis. PJK.
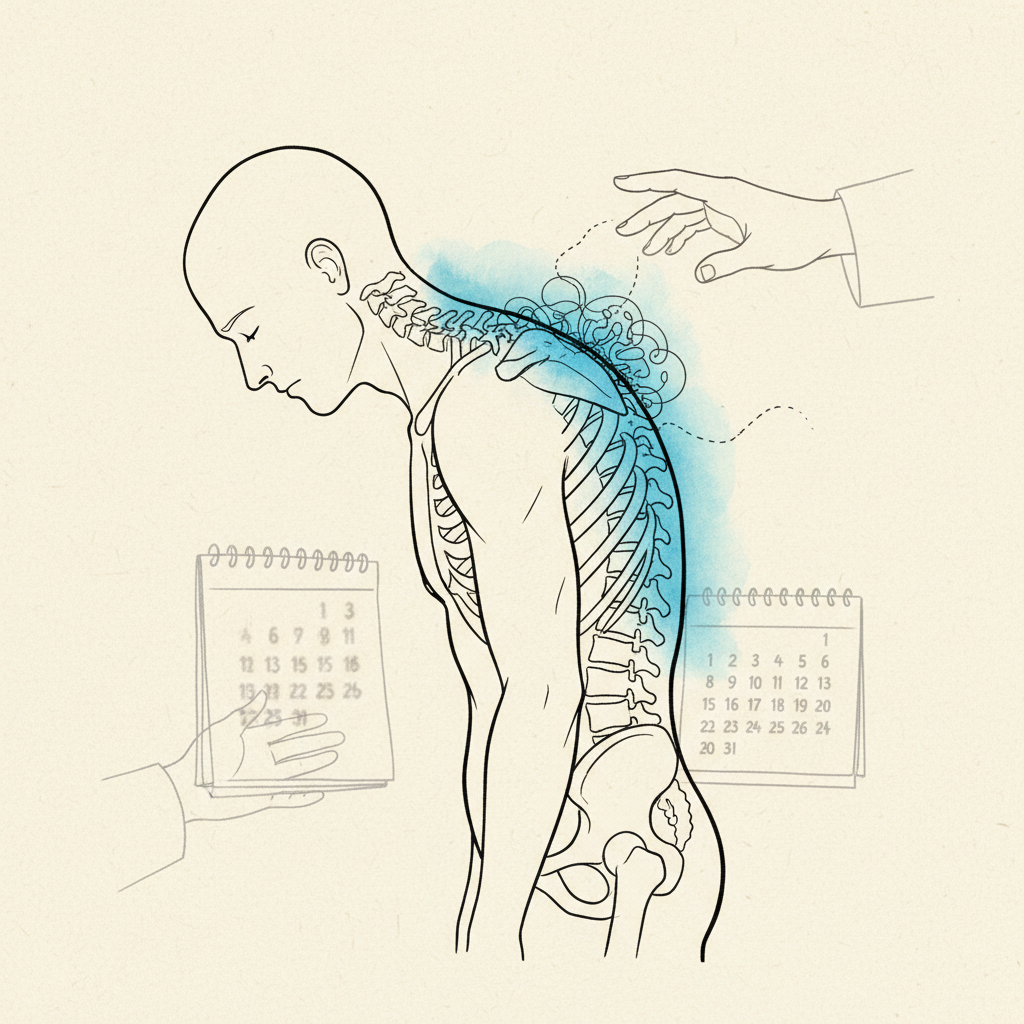
PJK is defined as an abnormal kyphotic angulation at the first mobile segment above the upper end of a spinal fusion construct. The segment that should be a smooth transition between the fused and unfused spine instead collapses forward. A new kyphotic angle develops where none existed before surgery.
The incidence is striking.
Hyun and colleagues published a systematic review in 2017 examining PJK after adult spinal deformity surgery. The reported incidence ranged from 20 to 40 percent (Hyun et al., 2017). Yagi and colleagues found similar rates in their 2012 analysis and identified the key risk factors: older age, osteoporosis, larger sagittal corrections, disruption of the posterior ligamentous complex during surgery, and the choice of upper instrumented vertebra (Yagi et al., 2012).
PJK is not a minor radiographic finding. It is the most common reason for revision surgery after adult spinal deformity correction. Smith and colleagues documented that revision rates following adult deformity surgery are substantial, with PJK and proximal junctional failure representing the leading indication for reoperation (Smith et al., 2012). Daniels and colleagues confirmed the scope of the problem in their 2019 analysis of complications following adult spinal deformity surgery, identifying junctional failure as one of the most clinically significant postoperative events (Daniels et al., 2019).
One in five. As many as two in five. Developing a new kyphotic deformity at the top of the construct that was supposed to resolve a deformity.
The surgical community knows this. The research is published. The risk factors are studied. The question that remains open is: what is actually driving the variability? Why do some patients develop PJK and others do not, even when the surgical and demographic factors are similar?
Proximal junctional kyphosis (PJK) is a kyphotic angulation that develops at the first mobile vertebral segment above a spinal fusion construct. It is the most clinically significant form of adjacent segment disease in adult deformity surgery. Hyun et al. (2017) conducted a systematic review reporting PJK incidence of 20-40% after adult spinal deformity surgery. Yagi et al. (2012) identified risk factors including older age, osteoporosis, larger sagittal corrections, disruption of the posterior ligamentous complex, and the specific upper instrumented vertebra chosen. PJK is the most common indication for revision surgery after adult deformity correction. Smith et al. (2012) documented substantial revision rates with PJK as the leading cause. Daniels et al. (2019) confirmed junctional failure as one of the most clinically significant complications following adult spinal deformity surgery. The clinical challenge is that surgical and demographic risk factors alone do not fully predict which patients will develop PJK.
The Mechanical Explanation
The standard surgical explanation for adjacent segment disease is mechanical. It is straightforward. It is supported by biomechanical studies. And it is real.
Fusion eliminates motion at the fused segments. The spine requires a certain total range of motion to perform daily activities. When the fused segments can no longer contribute, the adjacent segments must compensate. They move more. They bear more load per cycle. The increased loading accelerates the natural degenerative process at those levels.
This is Wolff’s law applied to the disc and facet joints. Tissues remodel in response to the loads placed on them. Excessive load accelerates degeneration. The transition segment bears the cost of the fusion’s rigidity.
In PJK specifically, the posterior ligamentous complex at the proximal junction is often disrupted during surgery. The paraspinal muscles are detached or weakened. The segment above the construct loses its posterior restraint. Gravity pulls the unsupported segment into kyphosis. Osteoporotic bone compounds the problem. The vertebral body itself can fracture under the new loading pattern.
This explanation accounts for much of the data. It explains why longer fusions produce more adjacent segment disease than shorter fusions. It explains why osteoporotic patients are at higher risk. It explains why disruption of the posterior ligamentous complex increases PJK incidence.
It is real. It is documented. It is incomplete.
It cannot explain why patients with nearly identical surgical constructs, bone density, and risk factor profiles show dramatically different outcomes. It cannot explain why some patients develop PJK within months while others never develop it. It cannot explain why the new deformity at the adjacent segment sometimes mirrors the pattern of the original deformity rather than following a generic degenerative pattern.
The mechanical explanation describes the vulnerability. It does not fully explain why the vulnerability is exploited in some patients and not others.
What the Generative Model Suggests
This section describes a hypothesis. It is not established surgical science. The components are individually supported by published research. The synthesis is our contribution. Read it as: “This is what the generative model of posture suggests as a contributing mechanism for the clinical variability in adjacent segment disease.”
Your brain maintains an internal model of your body called the body schema. This model, described by neurologist Jacques Paillard, operates below conscious awareness in the parietal cortex. It continuously generates your posture as a prediction (Paillard, 1999).
This is not metaphor. The predictive coding framework, formalized by Karl Friston as the free energy principle, establishes that the brain generates motor output as predictions based on its internal model. The body does not hold a position. The brain generates a position. Continuously. Automatically. Below awareness (Friston, 2010).
In scoliosis, in kyphosis, in any structural deformity that develops over time, the body schema is generating the curve as its best available prediction given the sensory data it has received. The curve is the output. The body schema is the generator.
Spinal fusion addresses the output. It mechanically prevents the curve from expressing at the fused segments. The hardware holds the vertebrae in their corrected position. The output is constrained.
But the generator was never addressed.
The body schema is still running the same prediction it was running before surgery. It is still generating the same motor commands. It is still organizing the body around the same pattern. The fused segments cannot respond to those commands. They are locked. But the unfused segments can respond. And they do.
The curve does not “migrate” to the adjacent segment because of mechanical overload alone. The curve migrates because the generator is still generating. The unfused segments are the only remaining territory where the generation can express itself. The pattern finds the path of least resistance. The transition zone, already mechanically vulnerable, becomes the new expression site for the same prediction that produced the original deformity.
This is a T2/T3 hypothesis. It has not been published as a unified framework in the surgical literature. But every component is established.
The body schema is established (Paillard, 1999). Predictive coding is established (Friston, 2010). The brain’s continuous generation of motor output as prediction is established. The fact that spinal fusion does not alter the neural model generating the deformity is a logical consequence of these established findings.
What this hypothesis adds is an explanation for the clinical variability that mechanical factors alone cannot account for. If adjacent segment disease were purely mechanical, the incidence should be more predictable from surgical and demographic variables. It is not. The variability suggests an additional factor. The generative model suggests that factor is the state of the neural prediction itself.
Patients whose body schema was strongly committed to the deformity pattern before surgery would be expected to show higher rates of adjacent segment disease. The generator is running the same program with higher confidence. It will find expression wherever it can. Patients whose neural model was less committed, or whose sensory environment shifted after surgery in ways that allowed the prediction to update, would show lower rates. The generator had less to express. Or it began generating something different.
This is testable in principle. It has not been tested. That is the boundary. We name it clearly.
The standard explanation for adjacent segment disease (ASD) after spinal fusion is biomechanical: fusion eliminates motion at the fused levels, increasing load on adjacent segments, which accelerates degeneration (Helgeson et al., 2010). This is supported by evidence. However, the variability in ASD incidence among patients with similar surgical and demographic profiles suggests additional contributing factors. The predictive coding framework (Friston, 2010) establishes that the brain generates posture as a continuous prediction from an internal model called the body schema (Paillard, 1999). In spinal deformity, the body schema generates the curve as its best prediction. Fusion constrains the output at the fused segments but does not alter the neural model generating the pattern. The generative model hypothesis suggests that the body schema continues generating the same prediction after surgery, and the unfused adjacent segments become the new expression site for the unchanged neural pattern. This hypothesis is consistent with the established components but has not been published as a unified framework in the surgical literature. It offers a potential explanation for why ASD variability exceeds what mechanical factors alone predict.
What This Means If You Are Considering Surgery
This is not an argument against spinal fusion. Fusion is sometimes necessary. Progressive neurological deficit. Intractable pain that has not responded to conservative care. Curves that threaten organ function. There are legitimate indications for surgery and surgeons who perform it with skill and precision.
What the generative model suggests is that the generator should be assessed before the output is mechanically locked.
If the body schema is generating the curve as a prediction, then the question before surgery is: can the prediction be updated? Can the sensory inputs to the body schema be changed in ways that alter the motor output? Can the nervous system’s safety state shift enough that the bracing pattern softens?
If it can, the curve may change without surgery. Not in every case. Not for every severity. But the question deserves to be asked before the answer is mechanically foreclosed.
If surgery proceeds, the generative model suggests that the neural prediction should still be addressed after fusion. The fused segments are stable. The unfused segments are now the vulnerable territory. If the body schema continues running the same prediction, those segments bear the full weight of the unchanged pattern plus the mechanical stress of the transition.
Addressing the generator after surgery means giving the nervous system new information. New sensory input to the body schema. Updated evidence about how the body is now organized. The prediction may update. The unfused segments may receive different instructions.
This is not a guarantee. It is a logical intervention based on the components described above. The generator was not addressed by surgery. Addressing it after surgery gives the adjacent segments better conditions than they would have if the old prediction continues unchecked.
The BrAIST study demonstrated that bracing reduces curve progression in adolescent idiopathic scoliosis (Weinstein et al., 2013). Bracing constrains the output. It is a mechanical intervention on a generated pattern. Fusion is the most definitive version of the same logic. Both constrain the output. Neither updates the generator. The question is whether updating the generator, before or after surgical constraint, changes the long-term trajectory of the adjacent segments.
If you are navigating this decision and want to understand the generative model in full, posturedojo.com is where we go deeper.
The question of prehabilitation before spinal fusion extends beyond traditional strength and flexibility preparation. The body schema, the brain’s internal model of the body maintained in the parietal cortex (Paillard, 1999), generates posture as a prediction (Friston, 2010). Spinal fusion mechanically constrains the output of this prediction at the fused levels. The generator itself is not altered by surgery. The generative model hypothesis suggests that updating the body schema before surgery, through sensory re-education and nervous system work that changes the inputs to the predictive model, may reduce the drive toward adjacent segment degeneration by altering the prediction that the unfused segments are executing. This is a hypothesis consistent with established neuroscience but not yet validated in the surgical outcome literature. Standard prehabilitation focuses on muscle conditioning and cardiovascular readiness. The generative model suggests an additional target: the neural prediction itself.
What This Means If You Already Had Surgery
If you have already had spinal fusion, the question is different. The hardware is in place. The fused segments are locked. What happened happened. The question now is: what can you do for the segments that are still mobile?
The answer is not reversal. Nobody is claiming that working with the nervous system after fusion will undo adjacent segment degeneration that has already occurred. Disc height that has been lost is lost. Facet arthropathy that has developed has developed.
The answer is conditions. Better conditions for the unfused segments going forward.
Three areas matter.
Sensory re-education. The body schema needs updated information about the body’s current organization. After fusion, the body is structurally different. The proprioceptive landscape has changed. But the body schema may still be running the pre-surgical prediction. Gentle sensory work that helps the brain map the new reality of the fused and unfused segments gives the model something accurate to work with. The prediction can update to reflect the body as it is now, not the body as it was before surgery.
Respiratory mechanics. The diaphragm and the ribcage are the hydraulic center of the torso. In patients with thoracolumbar fusion, respiratory mechanics are often compromised. Restoring diaphragmatic excursion and ribcage mobility in the unfused regions gives those segments hydraulic support. Pressure regulation. Not muscular bracing. The spine does not need to be held up by effort. It needs to be supported by pressure from the inside.
Transition zone awareness. The junctions between fused and unfused spine are the vulnerable territories. Building conscious awareness of these zones, not through force but through attention, gives the nervous system data it would not otherwise receive. The body schema maps what it pays attention to. Attention to the transition zones improves the resolution of the neural map at exactly the locations where PJK and adjacent segment disease develop.
None of this is a replacement for your surgical team’s guidance. It is an additional layer. The mechanical reality of your fusion is managed by your surgeon. The neural prediction running through the unfused segments is a different project. Both deserve attention.
Preventing adjacent segment disease (ASD) after spinal fusion is an active area of surgical research. Current strategies focus on surgical technique: preserving the posterior ligamentous complex, optimizing sagittal alignment, avoiding excessive correction, and choosing appropriate fusion endpoints (Yagi et al., 2012; Hyun et al., 2017). The generative model hypothesis suggests an additional avenue. Because the body schema continues generating motor predictions for the unfused segments after surgery (Paillard, 1999; Friston, 2010), post-surgical sensory re-education may help the neural model update to reflect the body’s new structural reality. This includes proprioceptive training for the transition zones between fused and unfused spine, respiratory mechanics to support hydraulic pressure regulation in the unfused thoracolumbar region, and body awareness practices that improve the neural map resolution at the vulnerable junctional segments. This approach is not a replacement for surgical follow-up care. It addresses a different system: the neural prediction that generates motor output for the mobile segments.
Post-surgical rehabilitation after spinal fusion typically focuses on wound healing, progressive mobility, and strengthening the muscles around the fusion construct. The generative model of posture suggests an additional rehabilitation target: the body schema. The body schema is the brain’s internal model of the body’s position and capabilities in space (Paillard, 1999). After fusion, the body’s structural reality has changed, but the body schema may still be running the pre-surgical prediction. Sensory re-education, including proprioceptive awareness of the transition zones between fused and unfused segments, respiratory mechanics training for the unfused thoracolumbar region, and gentle body awareness practices, may help the neural model update to reflect the body’s new organization. This does not replace standard post-surgical care. It addresses the neural prediction layer, which is a different system from the mechanical structure that surgery addressed. The goal is better conditions for the unfused segments by giving the body schema accurate, updated information about the body as it is now.
The Boundary
The mechanical explanation for adjacent segment disease is real. Increased loading. Accelerated degeneration. Disrupted ligamentous support. These are documented mechanisms with published evidence behind them.
The generative model adds a contributing hypothesis. Not a replacement. A contributor. The body schema was generating the original deformity. Fusion constrained the output but did not update the generator. The unfused segments became the expression site for the unchanged prediction. This is mechanistically consistent with established neuroscience. It has not been published as a unified framework in the surgical outcome literature. It is our synthesis.
We state it clearly so you can evaluate it clearly.
The mechanical factors are T1. Published. Replicated. Documented across multiple systematic reviews and long-term follow-up studies.
The generative model hypothesis is T2/T3. The components are T1. The synthesis is novel. It explains variability that the mechanical model alone does not. It generates testable predictions. It has not been tested in the surgical context.
Both can be true at the same time. Mechanical overload and an unchanged neural prediction are not competing explanations. They are layers of the same problem. The transition segment is mechanically vulnerable. And it is receiving instructions from a generator that was never updated. Both contribute. The question is how much each contributes, and whether addressing the generator changes the trajectory.
That question is open. We leave it open. The answer belongs to future research, not to certainty stated prematurely.
Related Reading
Scoliosis Treatment Without Surgery: The Complete Guide
Your Posture Is Generated. Here’s What That Changes.
How the Body Schema Controls Your Posture
85-Degree Scoliosis, No Surgery
Scoliosis Bracing: What It Actually Does
Sources
- Helgeson, M.D., Bevins, R.A., & Green, A.L. (2010). Adjacent segment degeneration after lumbar fusion: a systematic review. Spine Deformity. [T1]
Systematic review of adjacent segment disease. Radiographic ASD in 25-60% of patients at 10+ years. Symptomatic ASD in 5-25%. Established that ASD is the most common long-term complication following spinal fusion. - Cheh, G., Bridwell, K.H., Lenke, L.G., et al. (2007). Adjacent segment disease following lumbar/thoracolumbar fusion with pedicle screw instrumentation: a minimum 5-year follow-up. Spine, 32(20), 2253-2257. [T1]
Long-term follow-up of ASD after instrumented fusion. Documented radiographic degeneration in approximately one-third of patients, with a subset requiring revision surgery. - Ghiselli, G., Wang, J.C., Bhatia, N.N., et al. (2004). Adjacent segment degeneration in the lumbar spine. The Journal of Bone and Joint Surgery (American), 86(7), 1497-1503. [T1]
Ten-year incidence study. Adjacent segment disease developed in a significant proportion of lumbar fusion patients, establishing the long-term degenerative trajectory at transition segments. - Hyun, S.J., Lee, J., & Lee, B.H. (2017). Proximal junctional kyphosis and proximal junctional failure following adult spinal deformity surgery. Korean Journal of Spine, 14(4), 126-132. [T1]
Systematic review of PJK. Reported incidence of 20-40% following adult spinal deformity surgery. Identified PJK as the leading indication for revision surgery. - Yagi, M., King, A.B., & Boachie-Adjei, O. (2012). Incidence, risk factors, and natural course of proximal junctional kyphosis. Spine, 37(17), 1479-1489. [T1]
Risk factor analysis for PJK. Identified older age, osteoporosis, larger sagittal corrections, disruption of posterior ligamentous complex, and choice of upper instrumented vertebra as predictors. - Smith, J.S., Shaffrey, C.I., Glassman, S.D., et al. (2012). Risk-benefit assessment of surgery for adult scoliosis: an analysis based on patient age. Spine, 37(2), 125-132. [T1]
Revision rates following adult deformity surgery. Proximal junctional kyphosis and proximal junctional failure identified as leading indications for reoperation. - Daniels, A.H., Patel, A.A., & Vaccaro, A.R. (2019). Complications in adult spinal deformity surgery. Current Reviews in Musculoskeletal Medicine, 12(4), 468-474. [T1]
Comprehensive review of complications following adult spinal deformity surgery. Junctional failure identified as one of the most clinically significant postoperative events. - Weinstein, S.L., Dolan, L.A., Wright, J.G., & Dobbs, M.B. (2013). Effects of bracing in adolescents with idiopathic scoliosis. New England Journal of Medicine, 369(16), 1512-1521. [T1]
BrAIST trial. Demonstrated that bracing significantly reduces curve progression in adolescent idiopathic scoliosis. Establishes the mechanical constraint model: bracing constrains the output without altering the generator. - Paillard, J. (1999). Body schema and body image: A double dissociation in deafferented patients. In G.N. Gantchev et al. (Eds.), Motor Control, Today and Tomorrow. [T1]
Body schema as a neural model in the parietal cortex that generates postural predictions. Distinct from body image. Operates below conscious awareness. - Friston, K. (2010). The free-energy principle: a unified brain theory? Nature Reviews Neuroscience, 11(2), 127-138. [T1]
Predictive coding framework. The brain generates predictions and updates only when prediction error exceeds the model’s confidence. Posture is a generated prediction, not a held position.
Leave a Reply